About Denteric
Denteric is a clinical stage biotechnology company developing disease modifying therapies with the potential to treat inflammation caused by the gingipain toxins of the Porphyromonas gingivalis (Pg) bacterium. Our initial therapy is intended as an adjunct to scaling and root planing (SRP) to treat patients with severe disease or those refractory to the standard of care.
Denteric technology
We’re developing therapies that target gingipain toxins produced by P. gingivalis (Pg) including the proteolytic and adhesin domains that give the gingipains their virulence. GPV381, a therapeutic protein subunit vaccine, is our most advanced candidate. Administered by IM injection at the same time as SRP, this treatment is designed to:
Denteric Pipeline
GPV381
Denteric’s lead candidate GPV381 is an adjuvanted protein vaccine designed to treat severe or refractory periodontitis. It’s a disease-modifying therapy, changing the course of the disease rather than merely symptom control. Administered at the time of scaling and root planing, it is intended to induce neutralizing antibodies that can inhibit the gingipain toxin of P. gingivalis (Pg). Phase 1 trials are complete showing the vaccine to be safe and well tolerated.
Our pipeline includes the evaluation of GPV381 in the prevention or treatment of Alzheimer’s disease. This work is supported by funding from CUREator and Dementia Australia.
mRNA candidate
In addition to the lead protein vaccine GPV381, Denteric has also developed an mRNA vaccine candidate. The discovery program was supported by CRC-P funding from the Federal Government of Australia.
mAb Program
Currently at a discovery stage, Denteric is developing a range of monoclonal antibodies that can be used to support periodontal therapies.
Lead indication is severe periodontitis
P. gingivalis (Pg) has a key role in severe periodontitis, especially in the deep tooth pockets. By continuously releasing gingipain toxins into the pocket, it maintains an inflammatory state, leading to progressive tissue damage and bleeding. In healthy people, bacteria are readily cleared by the immune system, but Pg uses the gingipain toxins to shut down the local host immune response, providing itself with a protective environment in which to thrive.
Our therapy is intended as a treatment for those with severe periodontitis (predominantly stage 3 and 4 disease). Administered by health care professionals, as an adjunct to standard of care, the therapy is designed to activate the host’s immune system, inducing antibodies that can neutralize Pg gingipains both in the root pocket and the blood stream. This is intended to help the immune system to clear the toxins, stop inflammation and allow healing.
Most current therapies provide short-term benefit, often requiring the patient to attend frequent repeat maintenance sessions, in an attempt to control disease. GPV381 is designed to induce neutralising antibodies that will help to resolve the disease and provide continuous long-term protection. reduction of inflammation, restoration of immune control, and the healing and closing of periodontal pockets – true disease modification.
Unlike some therapies that are used to topically (locally) treat periodontal disease, we believe immunotherapy will have a longer-lived benefit.

Chronic diseases linked to Porphyromonas gingivalis
Pg is not only associated with severe periodontitis. There is a growing body of evidence identifying potential links between Pg and other diseases including Alzheimer’s disease, diabetes, cardio-vascular diseases, rheumatoid arthritis, and certain cancers. The chronic nature of periodontitis can expose the body to the gingipain toxins over many years and even decades. These toxins may travel throughout the body causing damage and inflammation. Furthermore, the ongoing inflammatory response in the mouth can spill over into the systemic circulation, accelerating aging and driving the chronic diseases.
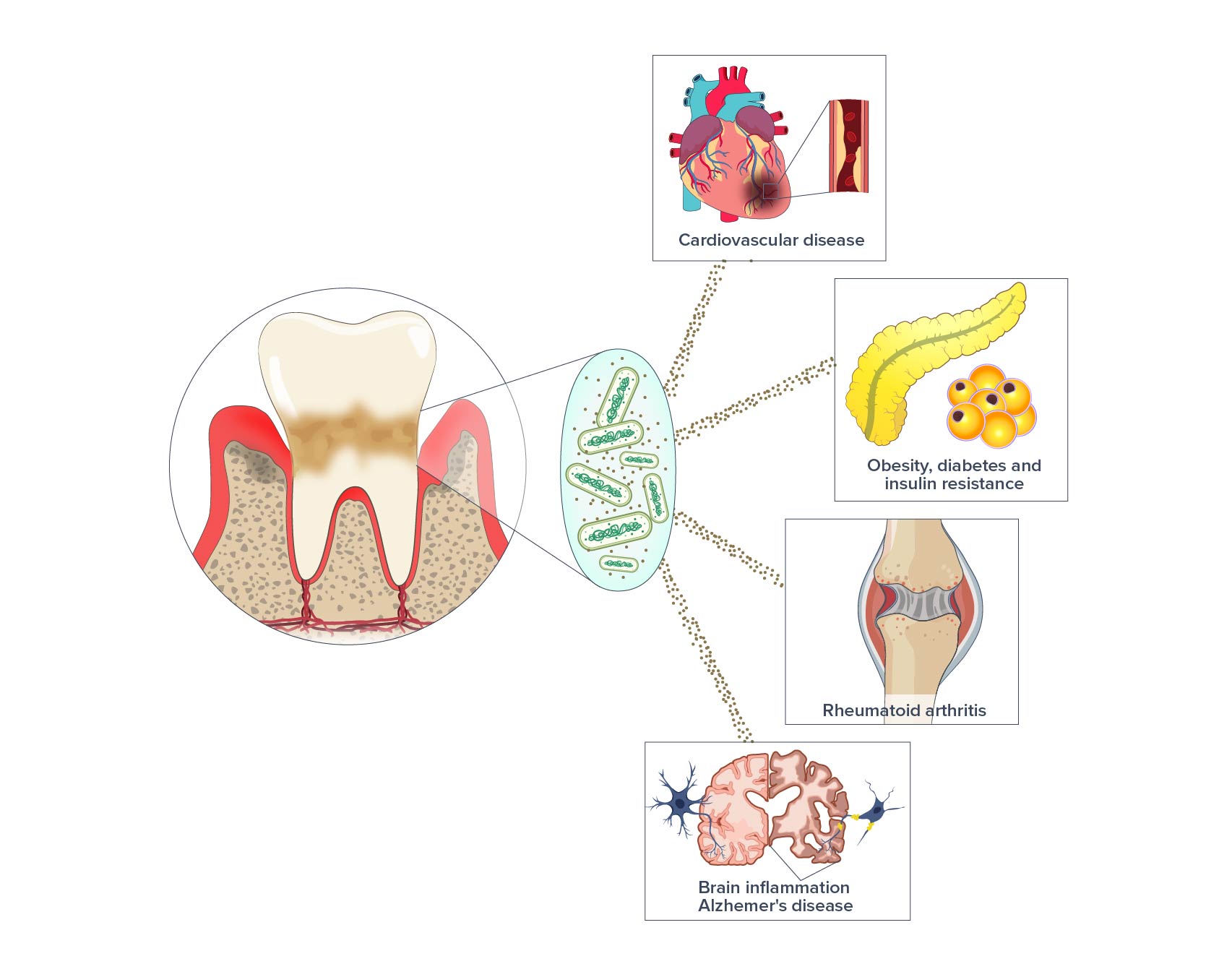
In time, we believe that we’ll be able to demonstrate the relevance of this approach to medical conditions associated with P. gingivalis such as Alzheimer’s disease.
Trial program
Please contact us on the form below if you have patients you think could benefit from joining the periodontal clinical trial program.
Our Investors
We’re a global team and headquartered in Australia. Our investors include:
Denteric People
Our team is filled with globally recognised experts in the field of Porphyromonas gingivalis research and immunotherapy development. Learn more about the people who are on a mission to develop the therapies that make severe periodontal disease a thing of the past.